How to Get Ozempic & GLP-1’s Covered by Insurance (or Save Big with Plum Health Direct Primary Care): The Ultimate Guide You Can’t Afford to Miss!
Plum Health
AUGUST 30, 2024
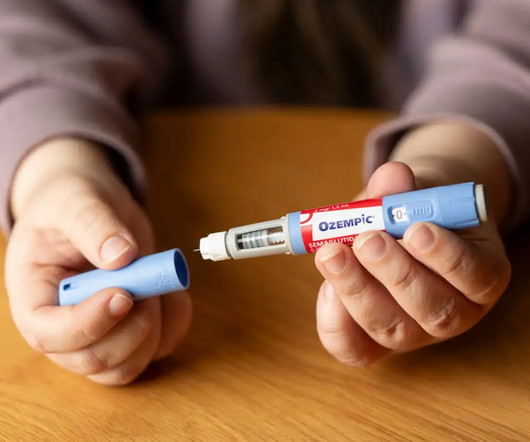
Mike to discuss affordable GLP-1 medication! Managing diabetes—especially as you get older—can be challenging physically, emotionally, and financially. These are groundbreaking medications that not only help regulate blood sugar but also support weight loss. It also slows the movement of food out of the stomach.
























Let's personalize your content