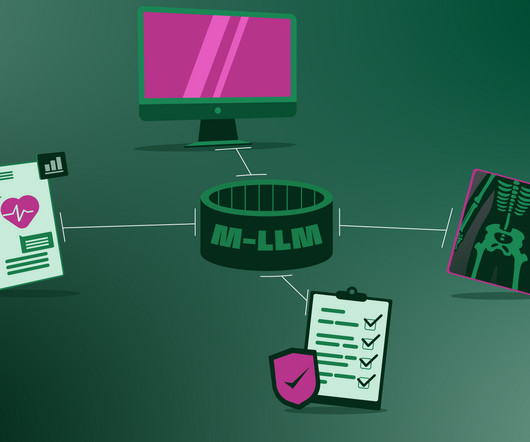
The Healthcare Vision of ChatGPT-4o and Multimodal LLMs
The Medical Futurist
JUNE 21, 2025
Although this revolution has been brewing for years, the past few months marked a major change , as algorithms finally moved out of the specialized labs and into our daily lives. These systems will considerably reduce the workload of – but not replace- human healthcare professionals.














Let's personalize your content