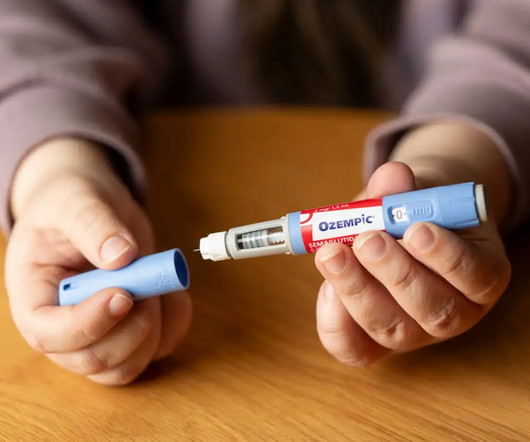
How to Get Ozempic & GLP-1’s Covered by Insurance (or Save Big with Plum Health Direct Primary Care): The Ultimate Guide You Can’t Afford to Miss!
Plum Health
AUGUST 30, 2024
Update: Click here to book a free 30 minute in-person consultation with Dr. Sharon or Dr. Mike to discuss affordable GLP-1 medication! Managing diabetes—especially as you get older—can be challenging physically, emotionally, and financially. With Plum Health, managing your diabetes becomes easier, more affordable, and less stressful.














Let's personalize your content