Researchers Propose Solutions to Improve GLP-1 RA Access
Physician's Weekly
JUNE 18, 2025
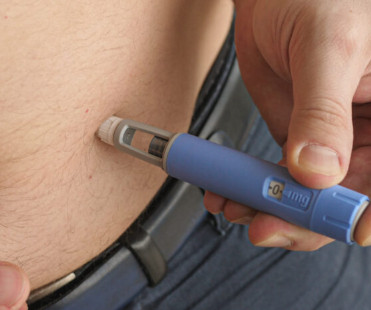
Cost-sharing and education may improve access to GLP-1 therapies for obesity, but systemic barriers remain, requiring broader policy and clinical reforms. The patient was enrolled in a cost-sharing program that reduced her out-of-pocket expenses by 70%, allowing her to access the medication.


































Let's personalize your content