We Have an LDT Proposed Rule!
FDA Law
SEPTEMBER 29, 2023
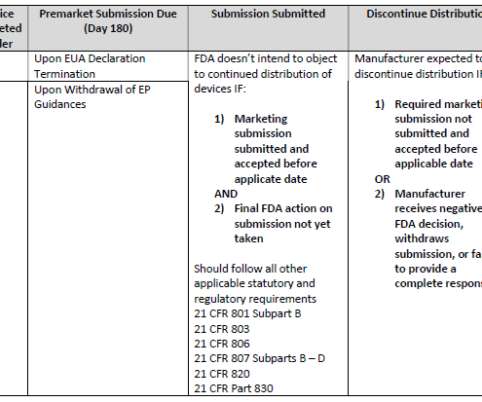
For more than 30 years, FDA has asserted that it has jurisdiction to regulate LDTs as medical devices and clinical laboratories as manufacturers. As we have blogged about extensively over the years, FDA has initiated, but not completed, many efforts through different means to create a regulatory framework for LDTs.










Let's personalize your content