The Rising Tide of Medical Waste: One Model for Improvement
My Green Doctor
MAY 3, 2025
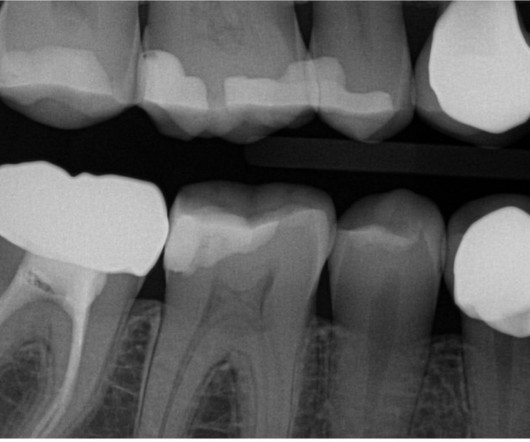
Innovative surgeries, novel pharmaceuticals, and state of the art technology are just a few of the driving forces that have aided the delivery of exceptional care to patients across all specialties. Dilating and anesthetic eye drops are used routinely to treat patients in most ophthalmology clinics. 3 “Each U.S.














Let's personalize your content